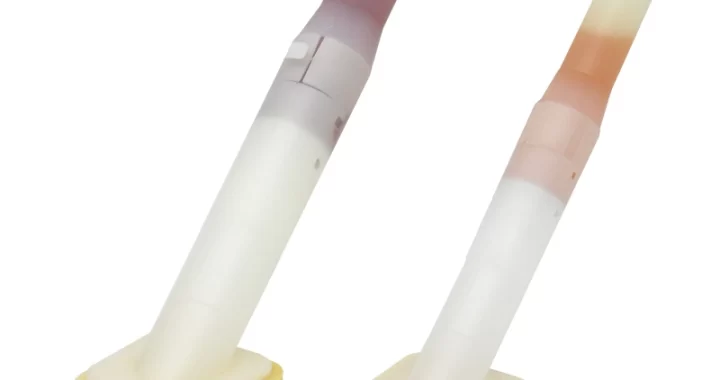
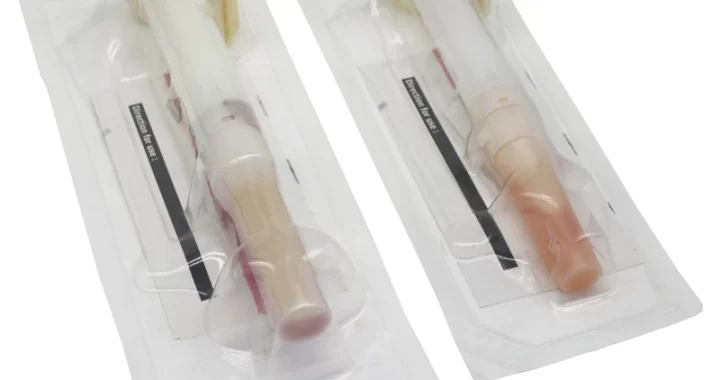
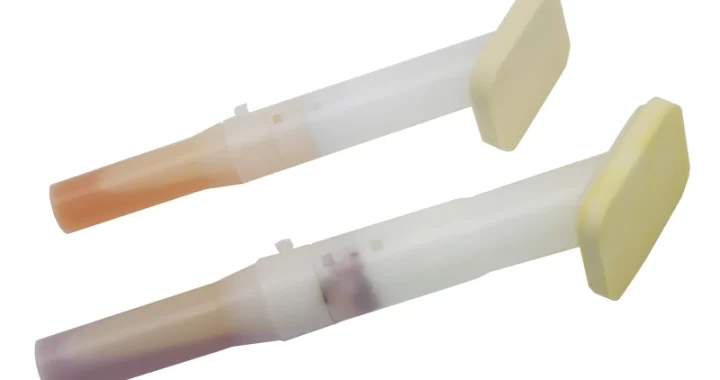
CHG disinfectant swabs are a medical-grade product designed for effective skin antisepsis. Their core features stem from the active ingredient—Chlorhexidine Gluconate (CHG)—combined with a convenient, ready-to-use applicator.
I. Core Efficacy (Driven by CHG Formula)
-
Broad-Spectrum Antimicrobial Action: Effective against a wide range of Gram-positive and Gram-negative bacteria, certain fungi, and some enveloped viruses.
-
Persistent Antimicrobial Effect: This is a defining feature. CHG binds to the skin to form a protective film, providing long-lasting activity (from several hours to days) that significantly reduces skin flora and inhibits regrowth.
-
Rapid Action: Begins to kill microorganisms quickly upon application.
II. Design & Convenience (Swab Applicator Advantages)
-
Individually Sterile-Packaged: Each swab is sealed separately, ensuring sterility, preventing contamination, and allowing for easy portability and storage.
-
Ready-to-Use Design: The swab stem is typically pre-filled with the CHG solution. It is activated by squeezing or breaking the stem (depending on design), instantly saturating the tip without needing separate disinfectants.
-
Precise & Easy Application: The swab format allows for targeted, controlled application with minimal dripping, ideal for small areas like injection or puncture sites.
-
Single-Use: Eliminates the risk of cross-contamination.
III. Application & User Benefits
-
Primary Use: Ideal for skin preparation before critical procedures, including:
-
Surgical site preparation
-
Intravenous (IV) catheter insertion and maintenance (e.g., PICC lines, central lines)
-
Blood culture collection and general injection sites
-
Cleaning around wound perimeters
-
-
Low Skin Irritation: Compared to other antiseptics like povidone-iodine, CHG (especially at common concentrations like 2%) is generally less irritating, less likely to cause allergic reactions, and does not stain the skin.
-
Non-Volatile: Unlike alcohol-based solutions, CHG does not evaporate quickly, allowing it to remain on the skin to provide persistent protection.
-
Visual Indicator (Common Feature): Many CHG swabs contain a colored dye (e.g., pink) to provide a clear visual guide of the disinfected area, ensuring complete and adequate coverage.
IV. Safety and Clinical Impact
-
Compliant with Medical Standards: Manufactured and sterilized (e.g., with Ethylene Oxide) to meet strict medical device requirements.
-
High Efficacy with a Favorable Safety Profile: CHG is proven to be safe and effective for skin antisepsis at recommended concentrations.
-
Proven Infection Reduction: The combination of rapid and persistent antimicrobial action is clinically proven to help reduce the risk of Surgical Site Infections (SSIs) and Catheter-Related Bloodstream Infections (CRBSIs).
Summary
In essence, CHG disinfectant swabs offer a powerful combination of:
-
Efficacy: Broad-spectrum + Persistent antimicrobial action (Core CHG benefit).
-
Convenience & Safety: Sterile, ready-to-use, single-use applicator (Swab form benefit).
-
User Experience: Precise application, low irritation, and visual confirmation.
They are primarily used for skin antisepsis prior to invasive medical procedures and for the maintenance of vascular access devices.