Description
CHG APPLICATOR Introduction
CHG Applicator is a disposable sterile medical device designed for professional skin antisepsis, taking Chlorhexidine Gluconate (CHG) as the core disinfectant component. Combined with humanized structural design, it achieves accurate, convenient and long-lasting skin disinfection, widely used in various clinical scenarios, and becomes an indispensable core tool in the modern medical infection control system, safeguarding the safety of medical operations and the recovery of patients.
Core Advantages
1. Core Ingredients for Long-lasting Bacteriostasis: Equipped with 2% Chlorhexidine Gluconate (CHG) combined with 70% Isopropyl Alcohol (IPA) optimized formula, complying with international authoritative standards such as FDA and ISO 13485. It has a broad-spectrum antibacterial effect, effectively killing gram-positive bacteria, gram-negative bacteria, fungi and some viruses, and even inhibiting drug-resistant bacteria such as MRSA and VRE. Unlike traditional alcohol and povidone-iodine, CHG can form an invisible antibacterial film on the skin surface, providing up to 24 hours of long-lasting bacteriostatic protection, effectively reducing the incidence of Surgical Site Infections (SSI) and Central Line-Associated Bloodstream Infections (CLABSI).
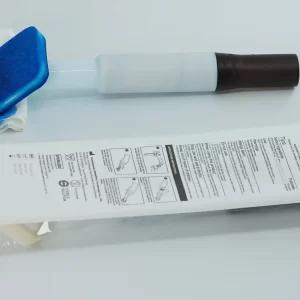
2. Humanized Design for Convenient Operation: Adopting an integrated design, consisting of a medical-grade ABS handle, a liquid storage capsule and a sponge applicator head. The handle is equipped with a convenient operation structure, and the standard dose of disinfectant can be released by rotating or pressing, without additional tools such as forceps, gauze and medicine bowls, which greatly simplifies the medical operation process and improves disinfection efficiency. The applicator head is made of 100PPI medical-grade open-cell polyurethane foam, which is soft, inert and hypoallergenic, with a debris loss of ≤1mg, fitting the skin without irritation, enabling uniform application and avoiding liquid leakage.
3. Sterile Protection for Safety and Control: The product is individually packaged in aluminum foil or blister, sterilized by gamma irradiation, and remains sterile before opening, effectively avoiding secondary pollution. Some products adopt triple sealing technology, with ultrasonic welding of the bottle mouth and double buckles of the liquid outlet, and the leakage rate is less than 0.1%, eliminating the risk of cross-infection, fully complying with the sterile operation specifications of the operating room. It is for single use only, discarded after use, no need for cleaning and disinfection, avoiding infection risks caused by repeated use, and latex-free, suitable for all groups of people.
4. Multiple Specifications for Flexible Application: Providing a full range of specifications, with liquid content covering 3ml, 5ml, 10.5ml, 26ml, etc., corresponding to different disinfection scenarios. The 3ml small round head is suitable for small-area disinfection such as venous puncture and PICC catheterization, and the 10.5ml and 26ml large round heads are suitable for preoperative large-area skin preparation and other scenarios. It also supports custom empty tubes, which can be filled with CHG solution of corresponding concentration according to needs, with stronger flexibility.
Application Scenarios
As a preferred clinical disinfection tool, CHG Applicator is widely used in multiple departments of medical institutions at all levels, recognized by authoritative institutions such as CDC, INS, SHEA and IDSA. The specific application scenarios include: preoperative preparation, invasive operations (PICC, CVC catheterization, hemodialysis vascular access preparation, lumbar puncture, biopsy, etc.), daily clinical disinfection (outpatient infusion, vaccination, wound dressing change, etc.), and emergency disinfection in intensive care units, emergency departments, obstetrics and radiology departments.
Quality Assurance
The products are produced in a 100,000-class clean workshop complying with GMP standards, equipped with a fully automatic filling production line. Strictly implement the three-level quality control system of raw material incoming inspection, production process monitoring and finished product factory inspection to ensure the consistency and stability of each batch of products. All products have obtained medical device registration certificates and passed ISO 13485 quality management system certification, with independent barcodes and validity date spray codes for quick batch traceability.
We are committed to providing high-quality, safe and reliable CHG applicators, adhering to the concept of “taking clinical needs as the guide”, continuously optimizing product performance, and providing customized solutions for medical institutions to help improve medical quality and protect patient safety.





Reviews
There are no reviews yet.